 27 because the Emergent plant has not received a green light from the agency. The rollout of the vaccine has been slow since the FDA awarded Johnson & Johnson an emergency use authorization on Feb. Three FDA officials, two investigators and a senior adviser signed off on the inspection. The 13-page report described nine separate problems, including a “failure to conduct thorough investigations into unexplained discrepancies,” facilities “not maintained in a clean and sanitary condition” and employees not being properly trained. An administration official noted that the nation's vaccination goals could be achieved without further Johnson & Johnson vaccine deliveries.Įmergent, a company with connections to former government officials, received at least $1.1 billion combined from the federal government and Johnson & Johnson last year to produce drug substance, the active ingredient in the vaccine. is approaching 200 million vaccinations, a milestone President Joe Biden highlighted in a speech Wednesday. 
this summer, a blow to vaccinating hard-to-reach populations that may face obstacles in getting a second shot as two other authorized vaccines require. The issues are not related to separate concerns that the Johnson & Johnson vaccine could be associated with exceedingly rare but potentially dangerous blood clots, the FDA clarified.īut the debacle raises serious questions about whether Johnson & Johnson can maintain its commitment to deliver 100 million of its one-shot doses to the U.S. No doses of the vaccine that have already been distributed and administered in the United States were contaminated, as they were sourced from the company’s plant in the Netherlands. "There is no assurance that other batches have not been subject to cross-contamination," reads the inspection document, known as an FDA Form 483. The inspection was triggered by an accident that ruined 15 million doses by cross-contaminating them with ingredients in the AstraZeneca vaccine.įDA inspectors found that plant operators did not do a thorough investigation into that incident, and that the integrity of other batches may also be compromised. 
company producing a crucial component of the Johnson & Johnson vaccine, and completed the inspection Tuesday. The agency launched an inspection on April 12 of the Baltimore plant of Emergent BioSolutions, the U.S. Meanwhile, the use of J&J's COVID-19 vaccine has been stalled in the US after the Food and Drug Administration (FDA) in May significantly restricted its use due to safety concerns.A contractor for Johnson & Johnson may have spoiled more COVID-19 vaccine doses than previously understood, according to a scathing Food and Drug Administration inspection report released Wednesday. In 2021, a manufacturing mishap at an Emergent plant in Baltimore rendered millions of Janssen COVID doses unusable. If the agreement is terminated, Emergent said J&J would owe it roughly $125 million to $420 million. J&J, as per an article in the Wall Street Journal, called that Emergent filing " false and misleading." The contract manufacturer added that J&J had failed to provide required forecasts for the amount of vaccines it needed and had wound down the agreement instead of fulfilling minimum requirements. 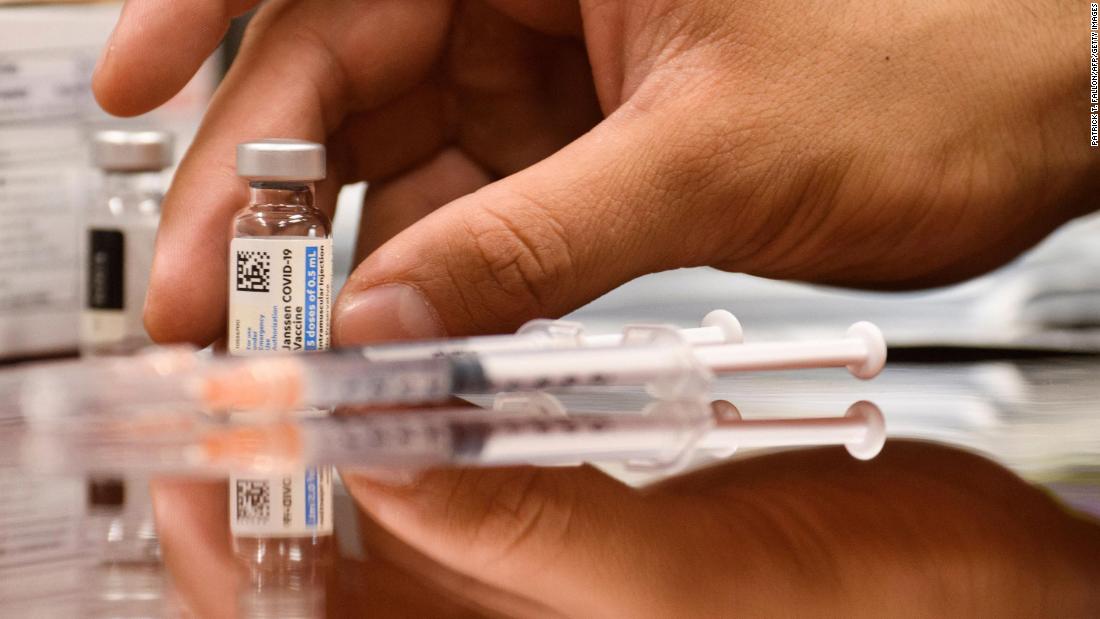
In a SEC filing yesterday, Emergent said that J&J breached an agreement by failing to buy the minimum quantity of COVID-19 vaccines made by the company. 
The termination notice, according to Reuters, comes about a week after J&J had initially informed Emergent of its intent to end the arrangement, with the drug maker citing what it termed the CDMO’s contract breaches, including failure by Emergent to perform its obligations in compliance with current good manufacturing practices (cGMP) or other applicable laws and regulations and its failure to supply Janssen with the product. Early July 2020 saw Emergent enter into a manufacturing services deal with Janssen Pharmaceuticals for large-scale drug substance manufacturing of J&J's investigational COVID-19 vaccine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
